In our In the Lab blog series, we catch up with researchers using CellScale systems.
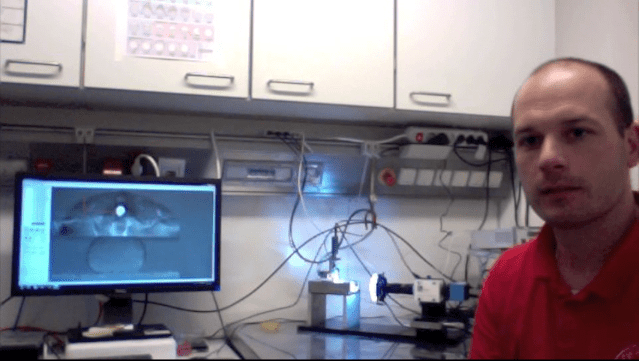
In this installment of the blog, Caleb Horst chats with Gabby Krenz of the Institute of Science and Technology in Austria. Gabby is completing research using the Microsquisher High Resolution Microscale Testing Machine.
Caleb Horst: For the last few years Gabby has been working in the lab with Professor C. P. Heisenberg, collaborating with CellScale on the development of the MicroSquisher testers. The work is ongoing in understanding the driving forces for cellular aggregation and organization.
Gabby, can you tell me a little bit more about the MicroSquisher and how you are using it for your research?
Gabby Krenz: I’m in the group of Carl-Philipp Heisenberg. We are interested in how you become from a one-celled embryo into a multi-cellular embryo. Of course, during development, different cell types are being induced and they need to find the proper place in the body of the developing organism. So one thought about this is that cells are finding their place in the organism based on the differences in the physical properties of these cells and these tissues. The MicroSquisher is a perfect device to probe these properties, not on a cellular level but more on a tissue level, as we are dealing with tissue-scale rearrangements.
CH: Can you walk me through a typical test and how you set it up and what you’re looking for and evaluating in the test?
GK: Of course. One of these physical properties I was just mentioning is the surface tension, which will be like comparing water and oil. If you shake two different liquids with different properties, like water and oil, the liquids will segregate, and this is based on the differences in their surface tensions. So, you can do a similar kind of experiment with different cell types in developing embryos, that is, in ectoderm tissue and mesoderm tissue, where you can also mix the cell types and they would separate into two different tissues. The driving force, as I mentioned, is the surface tension of these tissues, which can be measured by using homotypical aggregates (aggregates existing of only one cell type).
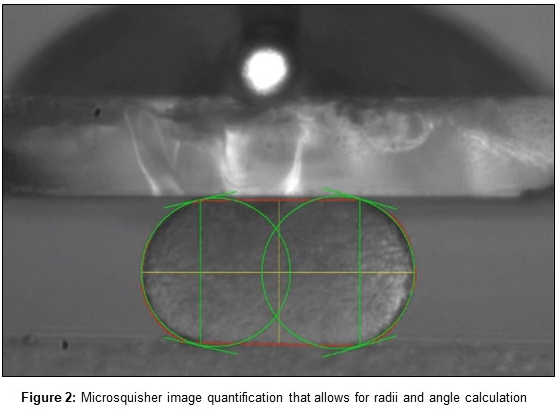
We isolate mesoderm tissues and let these small tissues round up into a nice sphere, which you can see here on the screen [Figure 1]. [And so] This is already a compressed aggregate, but what actually happens before compression is that the aggregate is round. The test we perform is to compress this aggregate until it’s flat, and then we simultaneously measure the force; how much force is needed to compress this tissue, but also how much force it takes to keep this tissue in this form.

CH: Can you give me some idea of the specimen sizes and the forces involved for the tests that you’re doing?
GK: Yes. Currently we are using specimen sizes of aggregates that are about 50 micrometers to 300 micrometers in diameter so we can vary the size of tissues. This is actually important because the surface tension is a measurement that should be independent of the size of the tissue or material that you’re probing because it’s a physical property. On average, we measure peak forces, which are on the low range for this machine but are between 1 and 3 microNewtons, and they would relax to a force that is 0.1 microNewtons to the minimum, depending on the tissue, of course, and the test that we apply.
CH: Can you tell me, has the MicroSquisher shed light on any new piece of information or led you to validate any assumptions that you started out with when you began this line of experimenting?
GK: So importantly, of course, things in development happen pretty fast and we are using zebrafish as a model organism which is known for its accessibility, but especially also because it has a very fast initial embryonic development. What we do specifically is follow the maturation of these tissues over time. Surface tensions are independent of size normally, but also a tissue should not change over time. We are working with biological material consistent cells that are adapting and changing to their environment, so one of the key features that we discovered is that over time as tissues are maturing or differentiating, these properties are changing over time. Then, comparing two different tissues on different and mutant tissues, one can look at the specific aspects of differentiation and see how aspects of differentiation influence changes in the physical properties of these tissues. Later, we need complementing experiments like, for instance, high confocal microscope imaging to confirm if cells also might rearrange in a different manner, or perform cell sorting experiments and in vitro experiments. But we can explain these experiments based on the measurements that we do here.
CH: Great, that’s very interesting work! Thanks for taking the time to talk with me Gabby, I really appreciate it. You’ve done a great job telling us about what you’re doing at the Institute for Science and Technology in Austria.
Watch the interview online: https://www.youtube.com/watch?v=5TjoRSw2hyE
CTA: Learn more about CellScale’s MicroTester or check out some of our customers’ publications.